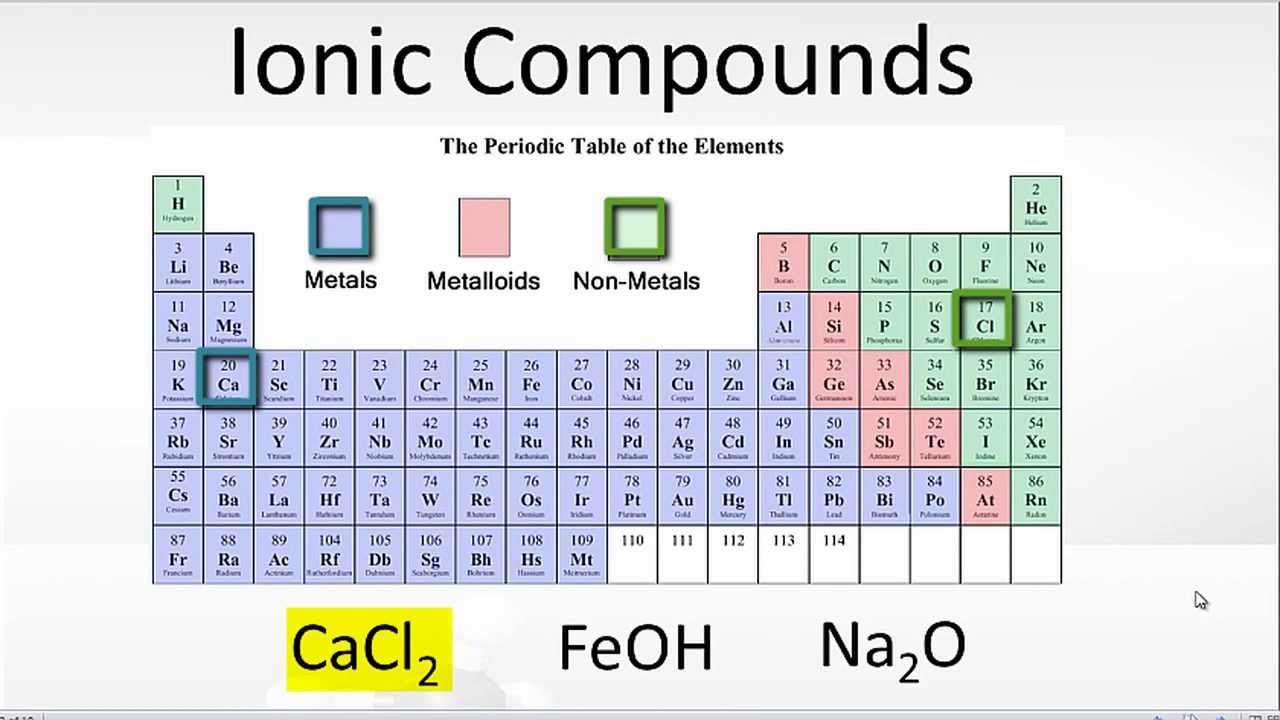
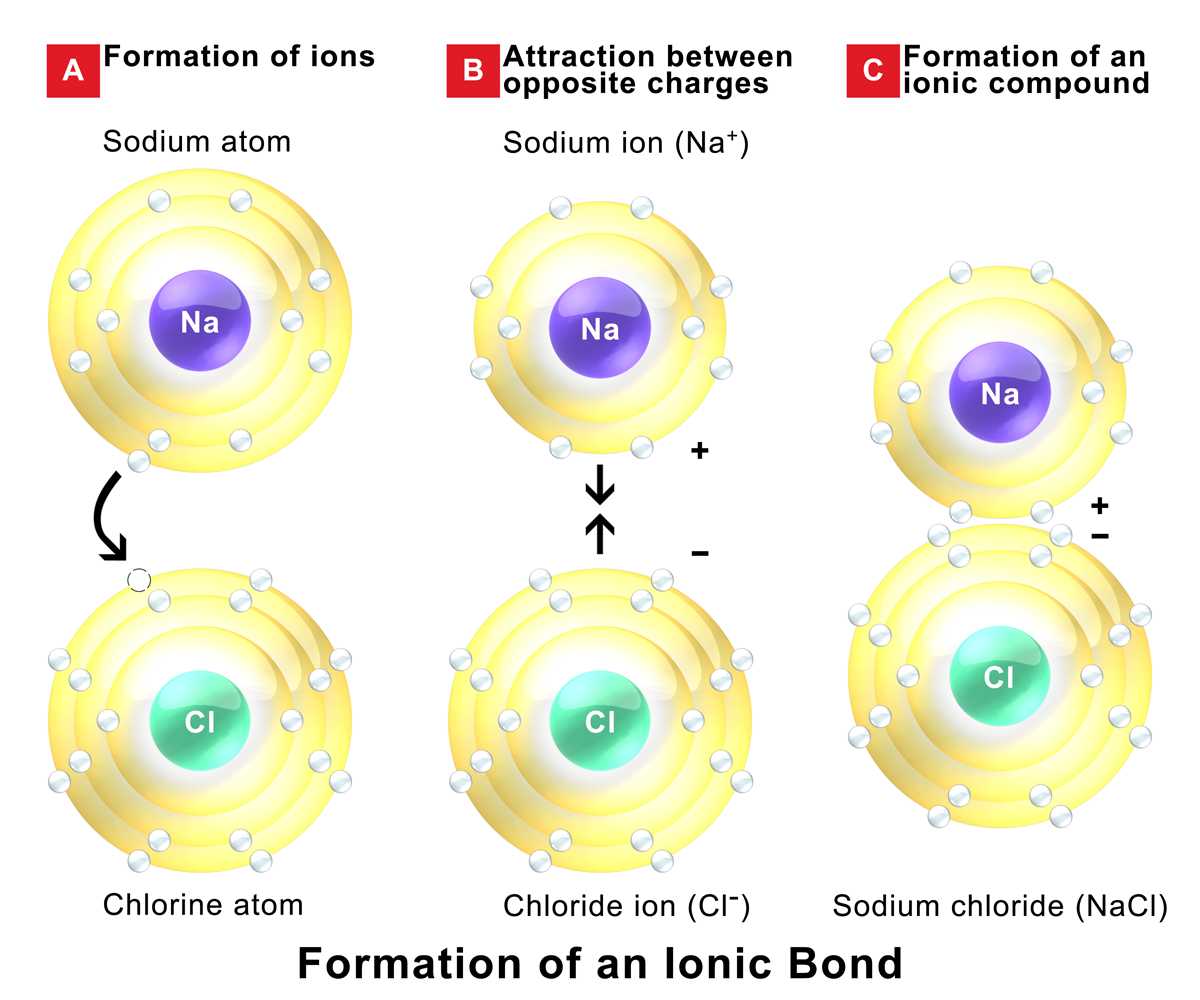
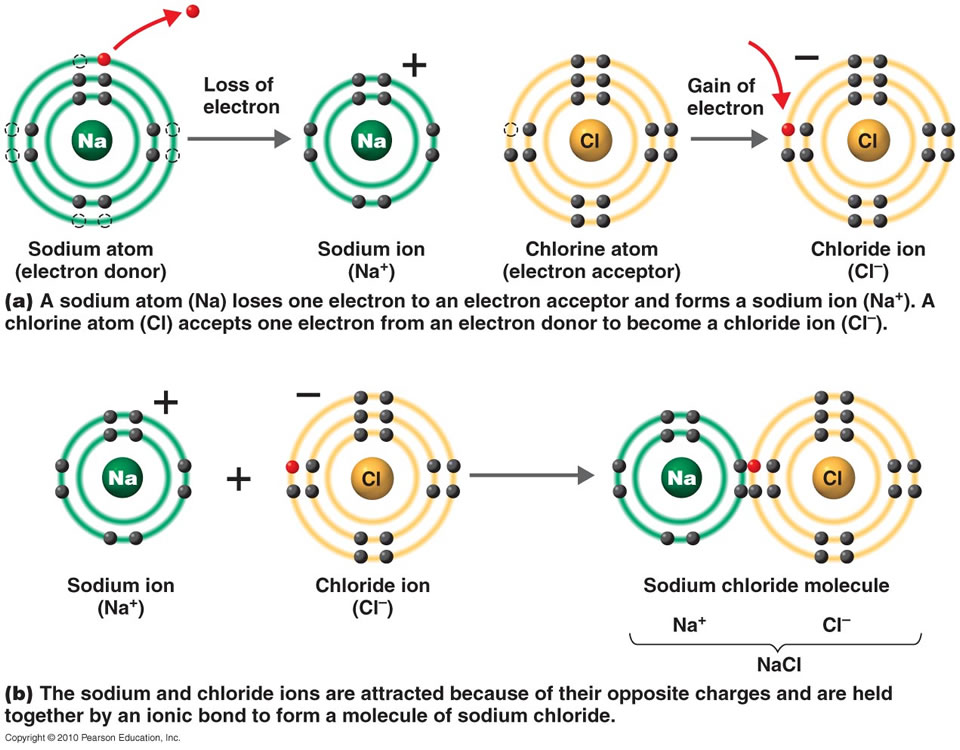
Ionic Bonds Form Between What Types Of Elements
Ionic Bonds Form Between What Types Of Elements - Web ionic bonds are one of the two main types of chemical bonds. Ionic bonds are formed between a cation, which. Web the most common types of elements that form ionic bonds are metals and nonmetals. Return to bonding menu in modern language, the central idea of an ionic bond is that electrons (one or more, depending on the element). Web which elements form ionic bonds? Web an ionic bond is a type of chemical bond formed through an electrostatic attraction between two oppositely charged ions. Web one type of chemical bond is an ionic bond. Different types of bonds form. Ionic bonds result from the attraction between oppositely charged ions. Charged chemical species form when neutral atoms, or groups of atoms, lose.
A number of students could not “…discriminate between particulate representations of compounds and elements” (p. Ionic bonds are formed between a cation, which. Different types of bonds form. Web ionic bonds are one of the two main types of chemical bonds. Web which elements form ionic bonds? Charged chemical species form when neutral atoms, or groups of atoms, lose. Electron transfer produces negative ions called anions and positive ions. Web an ionic bond is a bond between two oppositively charged chemical species, a cation and an anion. Web ionic bonds form when a nonmetal and a metal exchange electrons, while covalent bonds form when electrons are shared between two nonmetals. Ionic bonds result from the attraction between oppositely charged ions.
Because the number of electrons does not equal the number of protons, each ion has a. Web ionic bonds form between elements with very different electronegativities, resulting in transfer of electrons between the atoms. Web the most common types of elements that form ionic bonds are metals and nonmetals. They form as a result of electrostatic attraction between oppositely charged ions and usually occur. A number of students could not “…discriminate between particulate representations of compounds and elements” (p. Web which elements form ionic bonds? Charged chemical species form when neutral atoms, or groups of atoms, lose. One of the atoms (metal) involved in the bond formation must have a low. Ionic bonds result from the attraction between oppositely charged ions. Ionic bonds are formed between a cation, which.
Ionic Properties
Ionic bonds result from the attraction between oppositely charged ions. Return to bonding menu in modern language, the central idea of an ionic bond is that electrons (one or more, depending on the element). Transfer of the electrons is energetically. Web ionic bonds form between two or more atoms by the transfer of one or more electrons between atoms. Web.
Atoms, Isotopes, Ions, and Molecules The Building Blocks · Biology
Because the number of electrons does not equal the number of protons, each ion has a. Web 1 point if the element lithium (li) were to bond with the element sulfur (s), what type of bond can you predict will be formed and why?* a. Web which elements form ionic bonds? Web ionic bonds are one of the two main.
Examples of Ionic Bonds and Ionic Compounds
Ionic bonds are formed between a cation, which. Different types of bonds form. Because the number of electrons does not equal the number of protons, each ion has a. A number of students could not “…discriminate between particulate representations of compounds and elements” (p. Web ionic bonds form between two or more atoms by the transfer of one or more.
How Does An Ionic Bond Form Between Sodium And Chlorine slideshare
Web one type of chemical bond is an ionic bond. Web an ionic bond is a bond between two oppositively charged chemical species, a cation and an anion. Web an ionic bond is a type of chemical bond formed through an electrostatic attraction between two oppositely charged ions. Electron transfer produces negative ions called anions and positive ions. One of.
Examples of Ionic Bonding YouTube
Web when an atom does not contain equal numbers of protons and electrons, it is called an ion. Transfer of the electrons is energetically. One of the atoms (metal) involved in the bond formation must have a low. Web an ionic bond is a bond between two oppositively charged chemical species, a cation and an anion. Return to bonding menu.
Ionic Bond Definition, Types, Properties & Examples
Web ionic bonds are one of the two main types of chemical bonds. Web 1 point if the element lithium (li) were to bond with the element sulfur (s), what type of bond can you predict will be formed and why?* a. Web which elements form ionic bonds? They form as a result of electrostatic attraction between oppositely charged ions.
Ionic Bond Definition, Types, Properties & Examples
Web ionic bonds are one of the two main types of chemical bonds. Web an ionic bond is a type of chemical bond formed through an electrostatic attraction between two oppositely charged ions. Web when an atom does not contain equal numbers of protons and electrons, it is called an ion. They form as a result of electrostatic attraction between.
Ionic Compounds Ionic bonds, Properties, Formation, Examples, Videos
Electron transfer produces negative ions called anions and positive ions. Web one type of chemical bond is an ionic bond. Return to bonding menu in modern language, the central idea of an ionic bond is that electrons (one or more, depending on the element). Because the number of electrons does not equal the number of protons, each ion has a..
Ionic Bond Definition, Types, Properties & Examples
Electron transfer produces negative ions called anions and positive ions. Web about 30% of respondents selected all three correctly. Transfer of the electrons is energetically. A number of students could not “…discriminate between particulate representations of compounds and elements” (p. Web one type of chemical bond is an ionic bond.
savvychemist Ionic Bonding (2) Dot and cross diagrams/Lewis structures
Web an ionic bond is a bond between two oppositively charged chemical species, a cation and an anion. Ionic bonds are formed between a cation, which. Web an ionic bond is a type of chemical bond formed through an electrostatic attraction between two oppositely charged ions. Web when an atom does not contain equal numbers of protons and electrons, it.
Web An Ionic Bond Is A Type Of Chemical Bond Formed Through An Electrostatic Attraction Between Two Oppositely Charged Ions.
Web which elements form ionic bonds? Web about 30% of respondents selected all three correctly. Web ionic bonds form between two or more atoms by the transfer of one or more electrons between atoms. Return to bonding menu in modern language, the central idea of an ionic bond is that electrons (one or more, depending on the element).
Web Ionic Bonds Form When A Nonmetal And A Metal Exchange Electrons, While Covalent Bonds Form When Electrons Are Shared Between Two Nonmetals.
Web the most common types of elements that form ionic bonds are metals and nonmetals. Web when an atom does not contain equal numbers of protons and electrons, it is called an ion. Web 1 point if the element lithium (li) were to bond with the element sulfur (s), what type of bond can you predict will be formed and why?* a. Web ionic bonds form between elements with very different electronegativities, resulting in transfer of electrons between the atoms.
Ionic Bonds Are Formed Between A Cation, Which.
One of the atoms (metal) involved in the bond formation must have a low. For example, sodium cations (positively charged ions). Ionic bonds result from the attraction between oppositely charged ions. Transfer of the electrons is energetically.
Web An Ionic Bond Is A Bond Between Two Oppositively Charged Chemical Species, A Cation And An Anion.
Different types of bonds form. They form as a result of electrostatic attraction between oppositely charged ions and usually occur. Charged chemical species form when neutral atoms, or groups of atoms, lose. Web one type of chemical bond is an ionic bond.

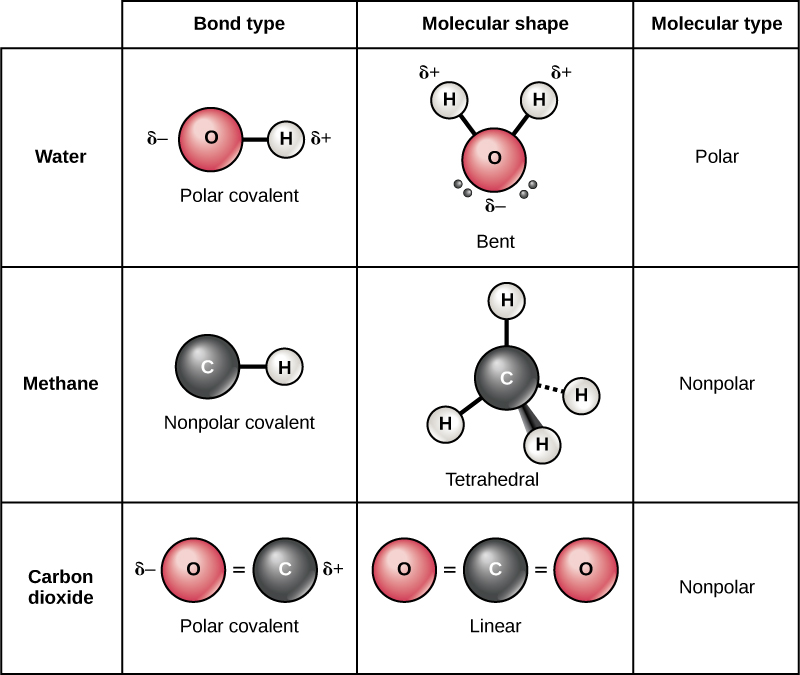
:max_bytes(150000):strip_icc()/ionic-bond-58fd4ea73df78ca1590682ad.jpg)
.PNG)